
It is always positive because the removal of an electron always requires that energy be put in (i.e., it is endothermic). IE is usually expressed in kJ/mol of atoms. For example, Sc and Ga both have three valence electrons, so the rapid increase in ionization energy occurs after the third ionization. Going down the group, ionization energy decreases and moving across a period ionization energy increases, so scandium has the highest ionization energy. Ionization energy (IE) is the amount of energy required to remove an electron from an atom in the gas phase: (14.5.3) A ( g) A + ( g) + e H I E. 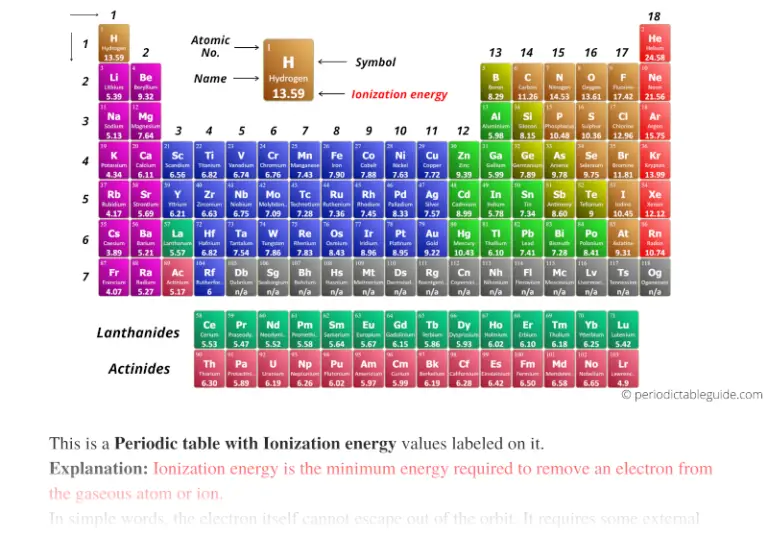
This jump corresponds to the removal of the core electrons, which are harder to remove than the valence electrons. The first ionization energy refers to the energy it takes to remove one electron from an atom. Choose the element with the greatest first ionization energy: Carbon or aluminum. As seen in the table below, there is a large increase in the ionization energies for each element. Thus, successive ionization energies for one element always increase. P A: (a) First ionization energy of P > first ionization energy of I. Likewise, removing an electron from a cation with a higher positive charge is more difficult than removing an electron from an ion with a lower charge. The first ionization energy for an element, X, is the energy required to form a cation with +1 charge: X(g) X+(g) +e IE1 (3.3.1) (3.3.1) X ( g) X + ( g) + e IE 1. Q: Choose the element with the higher first ionization energy in each pair.a. Study with Quizlet and memorize flashcards containing terms like Which element would release the most energy while adding an electron to a neutral atom in the gas phase, Selected properties of antimony (Sb) and iodine (I) are listed in the table below. Removing an electron from a cation is more difficult than removing an electron from a neutral atom because of the greater electrostatic attraction to the cation. To rank items as equivalent, overlap them. Rank from highest to lowest ionization energy. Krdo tv schedule tonight Sr Li Ca Se Sr < Ca < LiThe atom of. 
Choose the element with the higher ionization energy from each of the following pairs: Al or In Cl or Sb K or Ge S or Se. More specifically, this element will have its highest-energy electrons located in a 5p-orbital. The energy required to remove the third electron is the third ionization energy (IE 3) and so on. Arrange the following species in order of decreasing first ionization energy: Highest ionization energy to lowest Xe, Ar, Be, Ne. Remember that the first ionization energy (IE 1) is the energy required to remove the most loosely bound electron from a neutral atom and the second ionization energy (IE 2) is the energy required to remove the second most loosely bound electron.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
